Microelectrodes
Using Microelectrodes to Characterize Mass Transport in Ionomers and the Kinetics of the Hydrogen Oxidation and Oxygen Reduction Reactions
 One of the most important problems with current polymer-electrolyte fuel cells (PEFCs) is the cost caused by high Pt loadings in PEFC catalyst layers. Investigations into how to reduce these high Pt loadings are therefore critical. The Weber group uses microelectrodes as a model system to examine the interface between the Pt particles and ionomer in the catalyst layers. This allows for the mass transport resistances of oxygen and hydrogen in the ionomer film to be investigated. The kinetics of the hydrogen oxidation and oxygen reduction reactions can also be examined, depending on the reaction conditions. This will improve our fundamental knowledge of the Pt-ionomer interface and lead to the design of more efficient catalyst layers.
One of the most important problems with current polymer-electrolyte fuel cells (PEFCs) is the cost caused by high Pt loadings in PEFC catalyst layers. Investigations into how to reduce these high Pt loadings are therefore critical. The Weber group uses microelectrodes as a model system to examine the interface between the Pt particles and ionomer in the catalyst layers. This allows for the mass transport resistances of oxygen and hydrogen in the ionomer film to be investigated. The kinetics of the hydrogen oxidation and oxygen reduction reactions can also be examined, depending on the reaction conditions. This will improve our fundamental knowledge of the Pt-ionomer interface and lead to the design of more efficient catalyst layers.
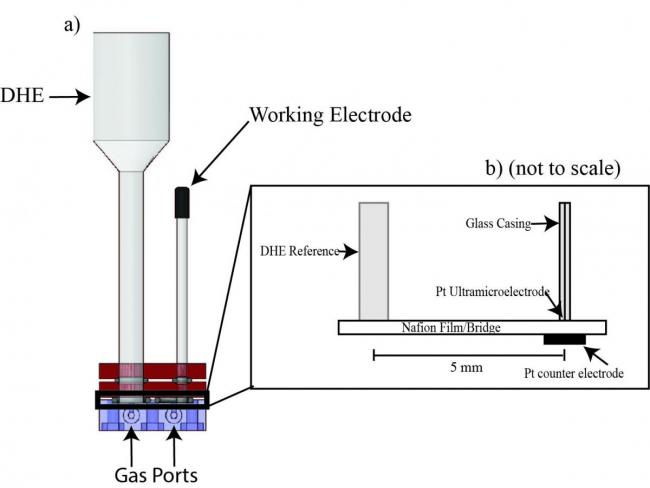
Figures at right: (Top) Schematic of microelectrode cell design. a) shows a cross-section from the side. The front and back of the UME cell each have a gas port for each chamber providing an inlet and outlet. b) shows a detailed layout of the components that make the cell circuit. (Bottom) Effect of mechanical pressure on both roughness factor and ORR current. In both cases, no discernible trend is observed, with little absolute change in values across the 2.76 MPa (400 psi) range. Roughness factor represents the ratio of electrochemically active surface area to geometric area and is a measure of the size of surface features on the Pt. The ORR current was determined using chronoamperometry.
Recent Publications:
Mass-Transport Resistances of Acid and Alkaline Ionomer Layers: A Microelectrode Study Part 1 – Microelectrode Development (ECS Fall 2019 Transaction, J. G. Petrovick, D. I. Kushner, M. Tesfaye, N. Danilovic, C. J. Radke, A. Z. Weber)